How Is Protein Powder Made? The Science Behind The Supplement


Every day, millions of scoops of protein powder are mixed into shakers, smoothies, and oatmeal bowls around the world. Yet very few of the people consuming them have any idea what it takes to produce what’s inside. If you’ve ever wondered exactly how protein powder is made, the answer lies in advanced filtration science, enzymatic chemistry, and tightly controlled manufacturing environments.
The raw ingredients – a bucket of milk, a pile of yellow peas – bear almost no resemblance to the fine, shelf-stable powder that ends up in a tub. That transformation is the result of a multi-stage industrial process, and its outcome depends almost entirely on the quality of the facility and the extraction methods used.
The Raw Ingredients: Sourcing the Foundation
Before a single gram of protein powder is produced, sourcing decisions are already shaping the final product.
Animal-Based Sources
The most common animal-derived proteins are whey and casein, both derived from cow’s milk. Raw milk is collected from dairy farms, transported under refrigeration, and immediately pasteurized – a heat treatment process that eliminates pathogenic bacteria without significantly altering the protein structure. What remains after pasteurization is the starting material for the separation process that follows.
Whey, specifically, is a byproduct of cheese production. When rennet is added to milk to coagulate the casein for cheesemaking, a thin liquid is left behind. That liquid is whey – and it’s surprisingly rich in complete protein, containing all nine essential amino acids.
Plant-Based Sources
Plant-based protein powders use a different class of raw materials entirely. The most common include:
- Yellow split peas – harvested, cleaned, and dehulled before processing
- Brown rice – milled and enzymatically treated to free the protein from the starch
- Soy – defatted and processed into isolate or concentrate form
- Hemp seeds – cold-pressed to remove the oil, leaving a protein-dense cake
Each plant source comes with its own amino acid profile and digestibility characteristics, which is why many plant-based formulas blend two or more sources to achieve a more complete profile. Regardless of origin, the raw material undergoes cleaning, sorting, and preliminary processing before entering the protein extraction phase.
The Extraction Process: How Protein Powder Is Made
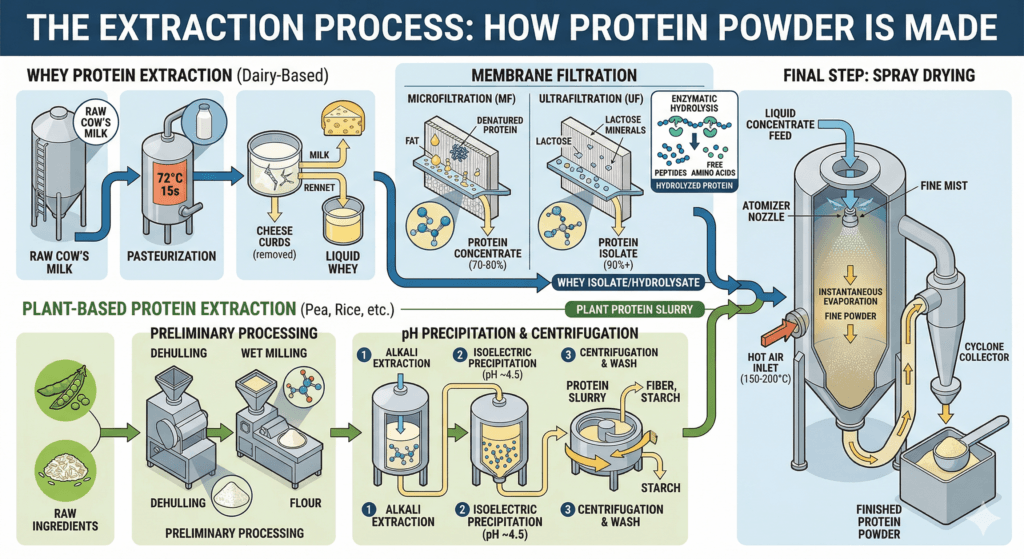
This is where the chemistry begins in earnest. The goal of the extraction phase is to isolate protein molecules from everything else in the raw material – fat, lactose, fiber, moisture – with minimal degradation of the protein’s nutritional structure.
Step 1: Pasteurization
For dairy-derived proteins, pasteurization is the first step. Raw milk or liquid whey is heated to a defined temperature for a defined period (typically 72°C for 15 seconds in continuous-flow pasteurization) to reduce microbial load before further processing.
Step 2: Separation
In whey processing, the liquid is run through centrifuges to separate fat globules from the protein-rich liquid. This produces a clarified whey stream that moves into the filtration stage.
Step 3: Microfiltration and Ultrafiltration
Microfiltration (MF)
Uses ceramic or polymer membranes with pore sizes typically in the 0.1–10 micron range. The liquid whey is pushed through these membranes under pressure. Larger molecules – fat and denatured proteins – are retained, while the smaller, intact protein molecules pass through. This is the primary method for producing whey protein concentrates (typically 70–80% protein by weight).
Ultrafiltration (UF)
Uses membranes with even smaller pore sizes (roughly 1-100 nanometers), applying higher pressure to remove lactose and mineral ions in addition to fats. This produces a higher-purity stream that, after further processing such as diafiltration, becomes whey protein isolate – often 90%+ protein by weight with minimal lactose.
Step 4: Ion Exchange (Optional)
Ion exchange chromatography uses the electrical charge of protein molecules to selectively bind them to a charged resin column, then release them using a salt or pH solution. The result is a very high-purity protein fraction. However, this method is sometimes criticized because the ionic binding process may denature certain minor protein fractions – such as lactoferrin and immunoglobulins – that are present in native whey but can be disrupted under the ion exchange conditions.
Step 5: Hydrolysis (For Hydrolyzed Proteins)
Enzymatic hydrolysis is the process by which specific proteolytic enzymes cleave the peptide bonds within protein chains, breaking long polypeptides into shorter chains called peptides, and in some cases, free amino acids. The resulting “hydrolyzed” or “pre-digested” protein is characterized by faster gastric transit and more rapid absorption in the small intestine compared to intact protein. Degree of hydrolysis (DH%) is measured and controlled to achieve specific molecular weight distributions in the final product.
For plant-based proteins, a similar sequence applies – wet milling or dry fractionation separates the protein from starch and fiber, followed by pH adjustment (acid precipitation or isoelectric precipitation), centrifugation, and wash cycles to produce a concentrated or isolated protein slurry.
Spray Drying, Blending, and Flavoring
At the end of the filtration phase, the protein exists as a high-moisture liquid – a slurry or concentrate that can contain 80–95% water. To convert this into a stable, scoopable powder, manufacturers use a process called spray drying.
Spray Drying
The protein-rich liquid is pumped through an atomizing nozzle at high pressure, which disperses it into a fine mist of microscopic droplets. These droplets are released into a large stainless-steel drying chamber through which heated air (typically 150-200°C at the inlet) circulates. The hot air contacts the droplets and evaporates the moisture almost instantaneously – in fractions of a second – before the protein droplets can be thermally damaged. What falls to the chamber floor is a fine, free-flowing powder with a moisture content typically below 5%.
The drying parameters – inlet temperature, outlet temperature, atomization pressure, and feed rate – are carefully calibrated to preserve the protein’s structural integrity while achieving the target particle size and bulk density.
Blending and Flavoring
The raw, unflavored powder is then moved into the formulation phase. This is where high-quality powder supplement manufacturing becomes critical. Industrial ribbon blenders or paddle mixers combine the base protein powder with:
- Natural or artificial flavors (e.g., cocoa powder, vanilla extract, natural flavor isolates)
- Sweeteners (sucralose, stevia, monk fruit, or combinations)
- Digestive enzymes such as protease, bromelain, or papain to improve protein breakdown
- Emulsifiers such as sunflower lecithin to improve mixability
- Stabilizers and flow agents such as silicon dioxide or xanthan gum
Blending time, speed, and sequence of ingredient addition are all process-controlled variables. A poorly blended batch produces uneven flavor distribution, clumping, or inconsistent amino acid profiles per serving – which is why precision at this stage matters.
Many of the trending supplement DTC brands differentiate themselves from the competition specifically during this blending and flavoring phase, investing in custom flavor systems, cleaner additive profiles, and proprietary enzyme blends that standard commodity powders don’t include.
Instantization
Some protein powders undergo a secondary agglomeration process – often called “instantization” – in which fine powder particles are re-moistened and dried to produce larger, more porous granules. This improves dispersibility in cold water and reduces the surface tension that causes clumping when the powder hits liquid.
Quality Assurance: Testing for Purity and Potency
Producing a protein powder that’s safe, accurately labeled, and consistent batch-to-batch requires a robust quality assurance framework. Reputable manufacturers operate under cGMP (current Good Manufacturing Practices) guidelines as defined by the FDA, which establish minimum standards for personnel, facilities, equipment, production controls, and record-keeping.
Third-Party Laboratory Testing
Independent certificate of analysis (COA) testing, conducted by accredited third-party labs, verifies several categories of product quality:
- Amino acid profile verification: Confirms that the stated protein content matches the actual molecular composition, and guards against “amino spiking” – the practice of adding cheap amino acids like glycine or taurine to inflate the total nitrogen reading.
- Heavy metal screening: Tests for lead, cadmium, arsenic, and mercury, particularly important for plant-based proteins derived from soil-grown crops.
- Microbial testing: Screens for total aerobic count, yeast, mold, coliforms, and pathogens such as Salmonella and Staphylococcus aureus.
- Allergen testing: Verifies absence of undeclared allergens – critical for products labeled “dairy-free” or “soy-free.”
Brands that pursue additional third-party certification marks (such as NSF Certified for Sport or Informed Sport) subject their products to even more stringent testing protocols, including screening for substances prohibited in athletic competition.
What It All Comes Down To
Turning raw agricultural materials into a functional, shelf-stable protein supplement is a genuine feat of applied food science – a marriage of agricultural sourcing, membrane separation chemistry, enzymatic engineering, and precision manufacturing. Every variable in the process, from the filtration membrane pore size to the spray dryer inlet temperature, has downstream consequences for protein purity, digestibility, flavor, and safety.
For entrepreneurs and brand owners, understanding this process is the first step toward making informed decisions about your formulation. The facility you partner with, the extraction method used for your protein base, and the blending controls in place during flavoring will determine the quality your customers experience in every scoop.
Ready to launch a scientifically backed protein formulation?
Partner with an expert manufacturing team that manages everything from raw ingredient sourcing to final packaging – so your product is built on the same science described here, not cut corners.
Plant-based and whey protein powders share some process steps – both go through filtration, drying, and blending – but the extraction methods differ significantly. Whey begins as a liquid dairy byproduct and is processed through membrane filtration. Plant proteins, such as pea or rice, are extracted from dry raw materials using wet milling, pH-based precipitation (adjusting the slurry to the protein’s isoelectric point to cause it to fall out of solution), and centrifugation. Plant proteins typically require additional processing to neutralize bitter or beany flavor compounds that are naturally present in the raw material.
The conversion from liquid to powder occurs through spray drying. The protein-rich liquid is atomized into a fine mist inside a large heated chamber. The moisture evaporates almost instantly on contact with the hot air, leaving behind dry protein particles that collect at the base of the chamber. The process takes milliseconds, which is fast enough to remove moisture without significant heat damage to the protein structure.
Mixability is addressed at two points in manufacturing. First, emulsifiers – most commonly sunflower or soy lecithin – are blended into the formulation to reduce the surface tension between the powder particles and water. Second, some powders undergo an instantization process in which the fine spray-dried particles are agglomerated into larger, more porous granules that disperse more readily in cold liquid. Particle size, bulk density, and moisture content all affect how a powder behaves when it hits a liquid.
Safety is established through a combination of cGMP-compliant manufacturing controls and multi-point quality testing. Facilities follow FDA-defined production standards that govern everything from equipment sanitation to allergen cross-contact prevention. Finished products are tested by independent laboratories for microbial contamination, heavy metals, and label accuracy before they are released for sale. These standards are regulatory requirements for supplement manufacturers operating in the U.S. market, and they are the baseline expectation for any product intended for daily use.
References:
NSF Certified for Sport – https://www.nsfsport.com/
Pasteurization – https://www.idfa.org/pasteurization
Enzymatic Hydrolysis – https://www.sciencedirect.com/topics/engineering/enzymatic-hydrolysis
COMMONLY ASKED QUESTIONS
Advertisement. This site offers health, wellness, fitness and nutritional information and is designed for educational purposes only. You should not rely on this information as a substitute for, nor does it replace, professional medical advice, diagnosis, or treatment. If you have any concerns or questions about your health, you should always consult with a physician or other health-care professional. Do not disregard, avoid or delay obtaining medical or health related advice from your health-care professional because of something you may have read on this site. The use of any information provided on this site is solely at your own risk.