How Is Creatine Monohydrate Made? The Manufacturing Process


When you’re building a supplement brand, knowing what goes into your products matters. Consumers are asking harder questions than ever – and “where does this come from?” is near the top of that list. For creatine monohydrate, one of the most studied performance ingredients on the market, the answer to that question starts in a laboratory, not a farm or a slaughterhouse.
| Creatine monohydrate is manufactured synthetically in cGMP-certified facilities by reacting two chemicals – sodium sarcosinate and cyanamide – with water in a pressurized reactor. The resulting crystals are purified, dried, and milled into a fine powder. No animal-derived ingredients are used at any stage of production. |
The Core Question: How Is Creatine Made Synthetically?
Creatine monohydrate – chemical formula C₄H₉N₃O₂ – is a naturally occurring compound found in skeletal muscle and synthesized in the human body from the amino acids arginine, glycine, and methionine. But the creatine in your supplement isn’t extracted from animal tissue or biosynthesized in a fermentation tank. Every gram of commercial creatine monohydrate is produced through direct chemical synthesis.
This is how creatine is manufactured synthetically: two industrial chemicals, sodium sarcosinate and cyanamide, are combined under controlled temperature and pressure to produce crude creatine. The reaction is well-understood, tightly controlled, and highly scalable – which is why synthetic manufacturing has become the universal standard for the ingredient.
The choice to synthesize rather than extract isn’t just practical. It’s also a quality decision. Synthetic production eliminates the batch-to-batch variability that comes with biological or animal-derived sources, and it allows manufacturers to test for and exclude contaminants at every stage. When done correctly, the result is a product with a purity profile that extraction methods simply can’t match at commercial scale.
Understanding how creatine is made gives formulators and brand owners a clearer picture of what “quality” actually means for this ingredient – and where the gaps between suppliers tend to appear.
The 4-Step Manufacturing Process of Creatine Monohydrate
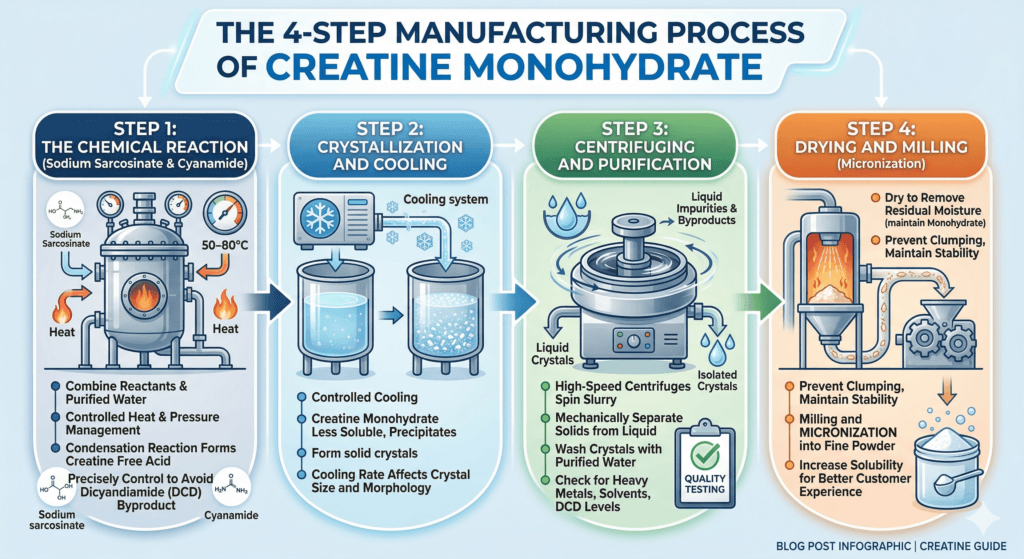
Step 1: The Chemical Reaction (Sodium Sarcosinate & Cyanamide)
The manufacturing process begins when sodium sarcosinate and cyanamide are combined in a pressurized reactor vessel along with purified water. Heat is applied – typically in the range of 50–80°C – while the reactor maintains controlled pressure throughout the reaction.
Under these conditions, the two compounds undergo a condensation reaction: cyanamide contributes a guanidino group to the sarcosinate backbone, forming creatine in its free acid form. The reaction is relatively fast by industrial chemistry standards, but precise temperature and pressure management is critical. Deviations at this stage could affect yield, purity, and the formation of unwanted byproducts – most notably dicyandiamide (DCD), a contaminant that quality testing is specifically designed to detect.
The output of this step is a hot aqueous solution containing dissolved creatine – essentially a liquid precursor to the final powder.
Step 2: Crystallization and Cooling
Once the chemical reaction is complete, the solution is cooled in a controlled manner. As the temperature drops, creatine monohydrate becomes less soluble in water and begins to precipitate out of the solution as solid crystals.
The rate and conditions of this cooling process directly influence crystal size and morphology. Manufacturers who invest in precise crystallization controls produce more uniform particle size distributions at this stage – a factor that has downstream implications for both purity and how efficiently the material behaves in the subsequent drying and milling steps.
The result is a slurry of crude creatine crystals suspended in residual liquid.
Step 3: Centrifuging and Purification
The crystal slurry is transferred to industrial centrifuges, which spin at high speed to mechanically separate the solid crystals from the surrounding liquid. The liquid phase – which contains dissolved impurities, unreacted starting materials, and reaction byproducts – is removed and discarded.
The isolated crystals are then washed with purified water to further reduce impurity levels. Depending on the manufacturing protocol, this wash cycle may be repeated multiple times. High-quality facilities also conduct in-process testing at this stage, checking for heavy metals, residual solvents, and DCD levels before the material moves forward.
This step is where a significant portion of the quality gap between manufacturers originates. Adequate centrifuging and washing time, combined with rigorous in-process testing, are non-negotiable for a finished ingredient that meets cGMP standards.
Step 4: Drying and Milling (Micronization)
The washed crystals are dried using industrial dryers – typically spray dryers or fluid bed dryers – to remove residual moisture. Moisture content matters for both stability and label compliance; creatine monohydrate must contain a specific ratio of water molecules in its crystal structure (monohydrate = one water molecule per creatine molecule) while remaining dry enough to prevent clumping during storage and handling.
Once the raw creatine crystals are formed, they must be milled into a fine, easily dissolvable powder. Partnering with an expert in powder supplement manufacturing ensures that the final product is properly micronized to prevent clumping and maximize solubility
Micronization – reducing particle size to the micron range – is what distinguishes a product that mixes cleanly into water from one that clumps, settles, or requires excessive agitation. This step is often underestimated by new formulators, but it has a direct impact on customer experience and product reviews.
Quality Control: What Separates Good Creatine From Bad?
Not all creatine monohydrate is created equal. Once you understand how creatine monohydrate is manufactured, it becomes clear that the process offers multiple points where quality can either be maintained or compromised.
- Dicyandiamide (DCD) contamination is the most commonly cited purity concern. DCD is a byproduct of the synthesis reaction that can persist into the finished ingredient if washing and centrifuging steps are inadequate. Reputable manufacturers test for DCD using HPLC methods and will provide certificates of analysis with quantified DCD levels – not just “not detected” language without a stated detection limit.
- Heavy metals including lead, arsenic, mercury, and cadmium can be introduced through raw material impurities or equipment contamination. Third-party ICP-MS testing is the standard for verifying heavy metal levels against USP or NSF limits.
- cGMP certification is a facility-level quality standard that governs everything from raw material intake to finished product release. Current Good Manufacturing Practices, as defined by the FDA for dietary supplements under 21 CFR Part 111, require documented procedures, validated equipment, trained personnel, and batch-level traceability. Ingredients sourcing from a cGMP-certified facility means those systems are in place and subject to audit – not just claimed on a spec sheet.
- Third-party testing – through organizations like NSF International, Informed Sport, or USP – adds an independent verification layer that goes beyond what a supplier’s in-house lab can provide. For brands positioning creatine as a clean, high-performance ingredient, third-party certification is increasingly a baseline expectation rather than a differentiator.
When evaluating suppliers, ask for full COAs, testing methodology, and facility audit documentation. The quality of your finished product is only as reliable as the quality controls upstream.
Is Synthetically Manufactured Creatine Vegan?
Yes – creatine monohydrate produced through chemical synthesis is 100% vegan.
The synthesis process uses sodium sarcosinate (derived from sarcosine, a plant- or petroleum-based chemical compound) and cyanamide (a nitrogen-carbon compound with no animal origin). No animal tissue, animal-derived enzymes, gelatin, or any other animal byproduct is involved at any stage of manufacturing.
This makes synthetically produced creatine monohydrate compatible with vegan and vegetarian diets – a meaningful distinction, since creatine is found naturally in animal muscle tissue and is often associated with meat-based nutrition in consumer perception. Clarifying that commercial creatine is made from chemical synthesis, not extracted from animals, is an important transparency point for brands marketing to plant-based audiences.
Brands should verify vegan status with their specific supplier, as facility practices (shared equipment, cross-contamination protocols) can affect certification eligibility even when the ingredient itself is animal-free.
Ready to Formulate?
Looking to formulate a high-purity creatine powder or pre-workout for your brand? Sourcing raw ingredients and ensuring proper micronization is critical to your product’s success. Explore our comprehensive powder supplement manufacturing services to see how Pronto can scale your business from R&D to final packaging.
SOURCING CREATINE SHOULDN’T BE THIS COMPLICATED.
Purity testing, micronization, cGMP compliance – we handle it all so you don’t have to. Partner with Pronto and give your brand a product worth standing behind.
Creatine monohydrate is manufactured synthetically by combining sodium sarcosinate and cyanamide in a pressurized reactor with water. The reaction produces crude creatine in solution, which is then crystallized, centrifuged, purified, dried, and milled into fine powder.
Yes. Synthetic creatine monohydrate is one of the most extensively studied ingredients in sports nutrition, with a strong safety record across decades of research. The key to safety is purity – a high-quality product from a cGMP-certified facility, tested for heavy metals and contaminants like DCD, is safe for consumption at established dosing levels.
Particle size – determined by the milling step – may influence dissolution rate, which can affect how quickly creatine enters solution in the gut. Properly micronized creatine dissolves more readily than coarsely milled material, but absorption of creatine monohydrate is generally high regardless of particle size when adequate water is consumed.
References:
Condensation Reaction – https://www.sciencedirect.com/topics/chemistry/condensation-reaction
21 CFR Part 111 – https://www.ecfr.gov/current/title-21/chapter-I/subchapter-B/part-111
NSF International – https://www.nsf.org
COMMONLY ASKED QUESTIONS
Advertisement. This site offers health, wellness, fitness and nutritional information and is designed for educational purposes only. You should not rely on this information as a substitute for, nor does it replace, professional medical advice, diagnosis, or treatment. If you have any concerns or questions about your health, you should always consult with a physician or other health-care professional. Do not disregard, avoid or delay obtaining medical or health related advice from your health-care professional because of something you may have read on this site. The use of any information provided on this site is solely at your own risk.