How to Choose a Supplement Manufacturing Partner in 2026


The US health and wellness market is on track to surpass $5 trillion in 2026, driven by sustained consumer demand for supplements targeting immunity, longevity, and performance. In a crowded marketplace, effective marketing may sell the first bottle – but product efficacy, purity, and label accuracy are what generate the repeat customer and the brand loyalty that sustains long-term growth.
The foundation of a successful supplement brand is not its label design or ad spend. It is its supply chain. For brand owners evaluating how to choose a supplement manufacturing partner, the process requires moving beyond unit cost to examine regulatory compliance, testing capabilities, and scalability. Every decision made at the manufacturing level either protects or exposes your brand.
This guide provides a structured, B2B framework for evaluating contract manufacturers – one that prioritizes documentation, data, and long-term operational fit.
Key Criteria at a Glance: Choosing a Supplement Manufacturer
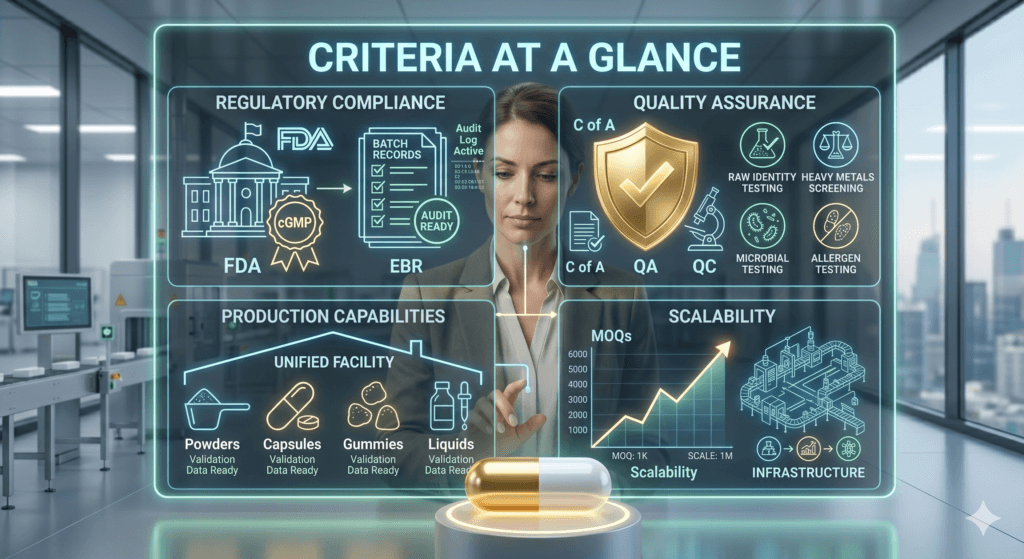
When establishing your product manufacturer selection criteria, prioritize these foundational pillars:
- Regulatory Compliance: Must operate an FDA-registered, cGMP-compliant facility utilizing digital supplement manufacturing compliance tools to ensure audit-ready batch records.
- Quality Assurance: Must perform raw material identity testing and final product screening for heavy metals, microbial contaminants, and allergens.
- Production Capabilities: Should offer the specific delivery formats (powders, capsules, gummies, liquids, etc.) required for your formulation under one roof.
- Scalability: Must provide viable Minimum Order Quantities (MOQs) with the infrastructure to scale production as your brand grows.
Establishing Your Product Manufacturer Selection Criteria
Defining rigorous product manufacturer selection criteria before you issue a single RFQ is one of the most consequential steps a brand can take. Without a structured evaluation framework, founders often default to selecting manufacturers based on price or proximity – two factors that rarely correlate with quality, compliance, or reliability.
The downstream costs of a poorly vetted manufacturing partner are severe: product recalls, FDA warning letters, supply chain disruptions, and legal liability that can erase years of brand equity. A checklist-driven approach removes subjectivity from the process and ensures that every candidate is evaluated against consistent standards.
Three overarching categories must be assessed:
- Regulatory standing – What certifications does the facility hold? What is its audit and inspection history with the FDA?
- Technological infrastructure – Does the facility rely on modern digital systems to manage quality, compliance, and traceability?
- Format capabilities – Can this manufacturer produce your specific product type, at the required volume and quality?
Each of these categories is explored in the sections that follow.
The Non-Negotiables: Regulatory Certifications and cGMP Compliance
Regulatory compliance is not optional. Under 21 CFR Part 111, the FDA mandates that all dietary supplement manufacturers operate under Current Good Manufacturing Practices (cGMP). These regulations govern everything from raw material receipt and testing to finished product labeling and distribution recordkeeping. A manufacturer that is not cGMP-compliant is not a viable manufacturing partner – it is a liability.
It is important to distinguish between two categories of regulatory standing. An FDA-registered facility has formally registered its operations with the FDA, as required by law. This registration does not constitute FDA approval of the products manufactured there. Third-party certifications – such as NSF International, USP (United States Pharmacopeia), or Informed Choice – go further. These programs require facilities to pass independent audits, validate manufacturing processes, and submit to ongoing product testing. Third-party certification is a meaningful signal of a manufacturer’s voluntary commitment to quality beyond the regulatory minimum.
Elite facilities maintain compliance not through manual paperwork, but through purpose-built supplement manufacturing compliance tools. These include:
- Electronic Quality Management Systems (eQMS) – Digital platforms that automate Standard Operating Procedure (SOP) management, deviation tracking, and corrective and preventive action (CAPA) workflows.
- Laboratory Information Management Systems (LIMS) – Software that tracks every aspect of laboratory testing from sample receipt through result approval, maintaining a complete, auditable data chain for each batch.
- Electronic Batch Record (EBR) Systems – Real-time digital logs that capture every manufacturing step, parameter, and sign-off as they occur, replacing paper-based records entirely.
Together, these systems automate batch record keeping, track ingredient lots across the supply chain, and ensure a facility can produce a complete compliance package – immediately, without scrambling – when an FDA audit occurs. When evaluating a manufacturing partner, ask directly whether these systems are in place. A vague or evasive answer is a disqualifying signal.
Evaluating Production Capabilities and Delivery Formats
A compliant manufacturer that cannot produce your specific product format is not a viable partner. The supplement industry spans a wide range of delivery formats, each with distinct manufacturing equipment, stability profiles, and regulatory considerations.
Common formats and their core considerations include:
- Powder blends – Require precise weighing, blending homogeneity validation, and moisture-controlled environments. Particle size consistency and flow properties are critical variables.
- Hard-shell capsules – Involve automated filling, capsule weight verification, and disintegration testing. Both vegetarian and gelatin shell options should be available.
- Softgels – A specialized process requiring dedicated encapsulation equipment suited to oil-based or lipid-soluble formulations.
- Gummies – Require gelatin or pectin cooking systems, mold deposition, coating, and moisture activity control to prevent microbial growth.
- Liquid formulations – Demand pH monitoring, preservative efficacy testing, and compatibility testing for container closure systems.
Beyond current product needs, brand owners must think about where their product line is going, not just where it starts. Brands that succeed with a single SKU almost always expand – adding complementary formats, seasonal products, or new delivery mechanisms. Managing multiple specialized contract manufacturers introduces compounding complexity: inconsistent quality standards, fragmented documentation, and escalating logistics costs.
Partnering with a facility that offers comprehensive, end-to-end supplement manufacturing services consolidates that complexity under one compliance umbrella, one set of quality standards, and one accountability structure.
When assessing a facility’s capabilities, request evidence of recent production runs in your specific format – not a list of formats they claim to offer. Ask for validation documentation and quality metrics from actual batches.
Ingredient Sourcing and Rigorous Quality Assurance
A top-tier manufacturer does not simply blend ingredients according to a formula. They serve as the first and most consequential line of defense in protecting product purity and potency. The journey of a raw material from supplier to finished product involves multiple testing checkpoints – each critical to label accuracy and consumer safety.
A defensible raw material quality program includes:
- Supplier qualification – Every ingredient vendor must be evaluated against defined criteria, including certifications, audit history, and quality documentation. Approved supplier lists should be maintained and reviewed on a regular cadence.
- Identity testing – Upon receipt, every raw material must undergo identity verification using validated analytical methods. Fourier-transform infrared spectroscopy (FTIR) and high-performance liquid chromatography (HPLC) are standard instruments for confirming that a delivered material is what the supplier claims it to be.
- Heavy metals screening – Ingredients are tested for lead, arsenic, cadmium, and mercury – non-negotiable given the bioaccumulation risks associated with contaminated botanical raw materials.
- Microbial testing – Finished products and select raw materials are screened for total aerobic count, yeast and mold, and the absence of specified pathogens including Salmonella and E. coli.
- Allergen controls – Facilities handling common allergens (tree nuts, soy, dairy, gluten) must implement verified cleaning protocols and maintain documented allergen matrices.
The manufacturer’s compliance tools should automatically generate a Certificate of Analysis (COA) for every production batch – documenting the testing performed, the specifications against which results are measured, and the pass/fail determination. Any manufacturer that cannot produce a current, third-party-verified COA on request is operating without adequate quality assurance infrastructure.
MOQs, Scalability, and Lead Times
A manufacturing partner must align with both the financial realities of your current stage and the growth trajectory you intend to pursue. Two business metrics govern this evaluation: Minimum Order Quantities (MOQs) and lead times.
MOQ is the smallest production run a manufacturer will accept. For early-stage brands, high MOQs create a difficult bind – tying up capital in inventory before market validation is complete. The right manufacturing partner offers accessible MOQs for initial product runs (typically 1,000 to 5,000 units depending on format) while possessing the production capacity to scale to hundreds of thousands or millions of units as demand warrants.
Lead time – the elapsed time from purchase order to finished goods receipt – is equally critical. It is driven by raw material procurement timelines, production scheduling, testing turnaround, and quality review. Ask any prospective partner to provide documented average lead times by format, and request references from current clients who can speak to on-time delivery performance.
Finally, evaluate scalability holistically. It is not just production capacity – it is whether the manufacturer’s quality systems, staffing, and procurement relationships can grow with your brand without service degradation.
3 Red Flags to Avoid in a Contract Manufacturer
During the vetting process, certain behaviors should trigger immediate disqualification.
- Refusal or inability to provide testing documentation. A manufacturer that cannot produce current Certificates of Analysis, does not operate electronic quality management systems, or struggles to answer basic questions about its testing protocols does not have the compliance infrastructure required under 21 CFR Part 111. This is a disqualifying deficiency – not a negotiating point.
- Pricing that falls significantly below market standards. Supplement manufacturing carries fixed costs: raw material testing, equipment validation, qualified laboratory staff, and compliance systems. A quote dramatically lower than competitive bids is not a bargain – it is a warning. Common cost-cutting practices at low-price facilities include “fairy dusting” (including an ingredient at sub-efficacious levels to justify a label claim), skipping identity testing on incoming raw materials, or sourcing from unverified suppliers. The resulting product may appear compliant but lacks the potency and purity consumers expect.
- Poor responsiveness during the quoting phase. The pre-sales interaction is a manufacturer’s best opportunity to demonstrate its professionalism and operational competence. Slow responses to RFQs, vague answers about certifications, inability to produce technical data sheets, or pressure to proceed without documentation review all predict how the relationship will function during production – and during a crisis.
Conclusion & Next Steps
Choosing a supplement manufacturing partner is one of the highest-stakes decisions a brand owner will make. The right partner is not simply a vendor – it is the operational foundation upon which your brand’s reputation for quality, safety, and consistency is built. Every certification, testing protocol, and quality system a manufacturer operates becomes an extension of the promise you make to your customers.
Evaluate thoroughly. Request documentation at every stage. Disqualify any partner that cannot demonstrate regulatory standing, technical testing capabilities, and the scalability to grow with your brand.
Ready to build a compliant, high-quality product line? Partner with a team that prioritizes transparency and precision at every step of the manufacturing process. Explore Pronto’s supplement manufacturing services to learn how the right manufacturing partner can accelerate your brand’s growth.
References
- Precedence Research. (2026). Health and Wellness Market Size, Share and Growth Report 2026–2035. U.S. market estimated at USD 5.02 trillion in 2026. https://www.precedenceresearch.com/health-and-wellness-market
- U.S. Food and Drug Administration. (2007). Current Good Manufacturing Practice in Manufacturing, Packaging, Labeling, or Holding Operations for Dietary Supplements. 21 CFR Part 111, 72 FR 34752. https://www.ecfr.gov/current/title-21/chapter-I/subchapter-B/part-111
- U.S. Food and Drug Administration. Current Good Manufacturing Practices (CGMPs) for Food and Dietary Supplements. https://www.fda.gov/food/guidance-regulation-food-and-dietary-supplements/current-good-manufacturing-practices-cgmps-food-and-dietary-supplements
- NSF International. NSF/ANSI 173: American National Standard – Product and Ingredient Certification Program. Annual audits, periodic retesting, and facility inspections required for certification. https://www.nsf.org/nutrition-wellness/product-and-ingredient-certification
- Capote Law Firm. (2025). Understanding cGMPs (21 CFR Part 111) for Dietary Supplements: Compliance & Best Practices. HPLC cited as standard method for active compound verification under 21 CFR Part 111. https://www.capotelawfirm.com/blog/understanding-cgmps-21-cfr-part-111
- Council for Responsible Nutrition. Dietary Supplement Health and Education Act (DSHEA). https://www.crnusa.org/regulation-legislation/fda-ftc-regulations/dietary-supplement-health-education-act-dshea
The most critical product manufacturer selection criteria are regulatory compliance, quality assurance capabilities, production format expertise, and operational scalability. A manufacturer must operate an FDA-registered facility under cGMP guidelines (21 CFR Part 111), conduct identity testing and contaminant screening on all raw materials and finished batches, produce your specific format with documented validation, and be capable of supporting your brand’s growth from initial run through high-volume production. Compliance documentation – including Certificates of Analysis and audit-ready batch records – must be readily accessible.
Supplement manufacturing compliance tools are digital systems that automate the documentation, tracking, and verification processes required under FDA cGMP regulations. Electronic Quality Management Systems (eQMS) manage SOPs, deviations, and corrective action workflows. Laboratory Information Management Systems (LIMS) maintain a complete, time-stamped data trail for every test performed on raw materials and finished products. Electronic Batch Records (EBR) capture manufacturing parameters in real time, eliminating the gaps common in paper-based systems. Together, these tools ensure that every production batch is traceable, every regulatory requirement is documented, and the facility can demonstrate compliance to auditors without delay.
The selection process differs meaningfully between the two. For custom formulas, the manufacturer must offer formulation development services, stability testing, and the R&D expertise to translate a concept into a validated, scalable formula. They must also be willing to protect your intellectual property through a Non-Disclosure Agreement and, ideally, a proprietary formula ownership clause. For white-label products, the evaluation focuses on the quality of existing formulations, the testing documentation supporting those formulas, and turnaround time for standard SKUs. In both cases, the core product manufacturer selection criteria – compliance, testing, format capabilities, and scalability – apply without exception.
No. The FDA does not approve dietary supplement manufacturers or the supplements they produce. Dietary supplements are regulated post-market under the Dietary Supplement Health and Education Act (DSHEA) of 1994 and the cGMP regulations codified in 21 CFR Part 111. Manufacturers are required to register their facility with the FDA and operate under cGMP standards, but this registration does not constitute FDA approval or endorsement of any product. The accurate description is an FDA-registered facility operating under cGMP guidelines – the phrase “FDA-approved supplement” is both inaccurate and inconsistent with FDA labeling and marketing requirements.
COMMONLY ASKED QUESTIONS
Advertisement. This site offers health, wellness, fitness and nutritional information and is designed for educational purposes only. You should not rely on this information as a substitute for, nor does it replace, professional medical advice, diagnosis, or treatment. If you have any concerns or questions about your health, you should always consult with a physician or other health-care professional. Do not disregard, avoid or delay obtaining medical or health related advice from your health-care professional because of something you may have read on this site. The use of any information provided on this site is solely at your own risk.